
The History of Automotive Metallurgy: From Steel to Aluminium
A chronological survey of how material selection in vehicle construction has evolved from Bessemer steel to advanced high-strength alloys over 130 years.
Read the full researchAn independent research publication examining how automotive metals respond to mechanical force at the molecular level — from crystal lattice theory to real-world structural behaviour.

The following comparative matrix presents key physical and mechanical properties of the three primary material groups used in modern vehicle architecture. Data sourced from peer-reviewed materials engineering standards.
| Property | Mild Steel (SPCC) | High-Strength Steel (HSS) | Aluminium Alloy (6061-T6) | Magnesium Alloy (AZ31B) |
|---|---|---|---|---|
| Density (g/cm³) | 7.85 | 7.85 | 2.70 | 1.77 |
| Tensile Strength (MPa) | 270–410 | 550–1,500 | 310 | 260 |
| Yield Strength (MPa) | 140–280 | 350–1,200 | 276 | 200 |
| Young's Modulus (GPa) | 200 | 200–210 | 69 | 45 |
| Elongation at Break (%) | 28–38 | 8–25 | 12–17 | 10–15 |
| Hardness (Brinell, HB) | 95–130 | 160–450 | 95 | 49–73 |
| Thermal Conductivity (W/m·K) | 50 | 48–52 | 167 | 96 |
| Corrosion Resistance | Low (requires coating) | Low–Moderate | High (native oxide layer) | Moderate (surface treatment needed) |
| Primary Automotive Application | Door skins, floor panels, bonnets | Structural pillars, crash rails | Bonnets, door structures, chassis | Dashboard frames, seat structures |
| Relative Weldability | Excellent | Good (requires pre-heat) | Moderate (MIG/TIG required) | Poor (specialist equipment) |
At the core of metallic behaviour is the crystalline lattice — a three-dimensional arrangement of atoms held in equilibrium by electromagnetic bonds. When a force is applied to a metal component, these atomic bonds are temporarily stretched. Provided the stress does not exceed the material's elastic limit, the lattice returns to its original configuration upon removal of the load. This phenomenon is what engineers refer to as elastic recovery or, colloquially, molecular memory.
The precision of this recovery depends on several interrelated variables: grain size, dislocation density, alloy composition, and the rate at which the stress was applied. Fine-grained steels produced through controlled rolling processes typically exhibit superior elastic recovery compared to coarse-grained equivalents. The presence of carbon atoms in the iron lattice creates what metallurgists call interstitial solid solutions — regions of heightened lattice tension that resist deformation up to a well-defined threshold.
Understanding molecular memory is fundamental not only to engineering but also to the science of failure prediction. When the elastic limit is surpassed, dislocations multiply and migrate, creating permanent changes in grain orientation. These changes, invisible to the naked eye, alter the material's future stress response — effectively recording the history of applied forces in the metal's microstructure.

Pure metals in isolation rarely meet the complex demands of structural automotive engineering. Through the deliberate addition of secondary elements — a process fundamental to physical metallurgy — manufacturers engineer alloys whose properties far exceed those of any constituent material acting alone.
Increases hardness and tensile strength through interstitial lattice occupation. Higher carbon content shifts steel from mild to high-carbon grade, reducing ductility proportionally.
Refines grain structure, increases hardenability, and prevents hot-shortness by combining with sulphur. Present in nearly all engineering steels at 0.3–1.6 wt%.
Acts as a deoxidiser during steel production and strengthens the ferrite matrix through solid-solution hardening. Also critical in aluminium alloys of the 6xxx series.
Forms a passive chromium-oxide surface layer, providing corrosion resistance. Increases hardenability and carbide formation in tool and stainless steel grades.
Improves toughness at low temperatures by stabilising the austenitic phase. Widely used in high-performance automotive alloys requiring thermal resistance.
The lightest structural metal used commercially. In aluminium alloys (5xxx, 6xxx series), magnesium enhances strength while preserving formability.

Temperature exerts a profound influence on the mechanical properties of automotive metals. As thermal energy increases, atomic vibration amplitudes widen, reducing the net binding force between atoms and lowering the material's yield strength. This relationship is not merely academic — it governs everything from the performance of structural panels during extreme driving conditions to the behaviour of metal during manufacturing processes such as hot stamping and roll forming.
Steel, at temperatures above its recrystallisation threshold (approximately 450–720°C depending on grade), undergoes grain refinement when mechanically worked — a process exploited in controlled rolling mills to produce ultra-high-strength panels. Aluminium, with its significantly lower melting point of 660°C, requires tighter thermal management during forming operations to prevent localised softening.

Determining the precise elastic limit of a metal is among the most important measurements in structural engineering. Unlike the ultimate tensile strength — the point at which a material fractures — the yield strength defines the boundary between recoverable deformation and permanent structural change. Below this threshold, the material behaves as a spring; above it, as plastic clay.
Modern engineering laboratories employ several standardised methodologies to characterise yield behaviour with high precision. Each technique offers different advantages depending on the material, loading rate, and intended application.

Peer-reviewed and editorial articles exploring the intersection of physical metallurgy and automotive engineering science.

A chronological survey of how material selection in vehicle construction has evolved from Bessemer steel to advanced high-strength alloys over 130 years.
Read the full research
An in-depth examination of how metallic crystal lattices encode deformation history and the physical mechanisms governing elastic recovery.
Read the full research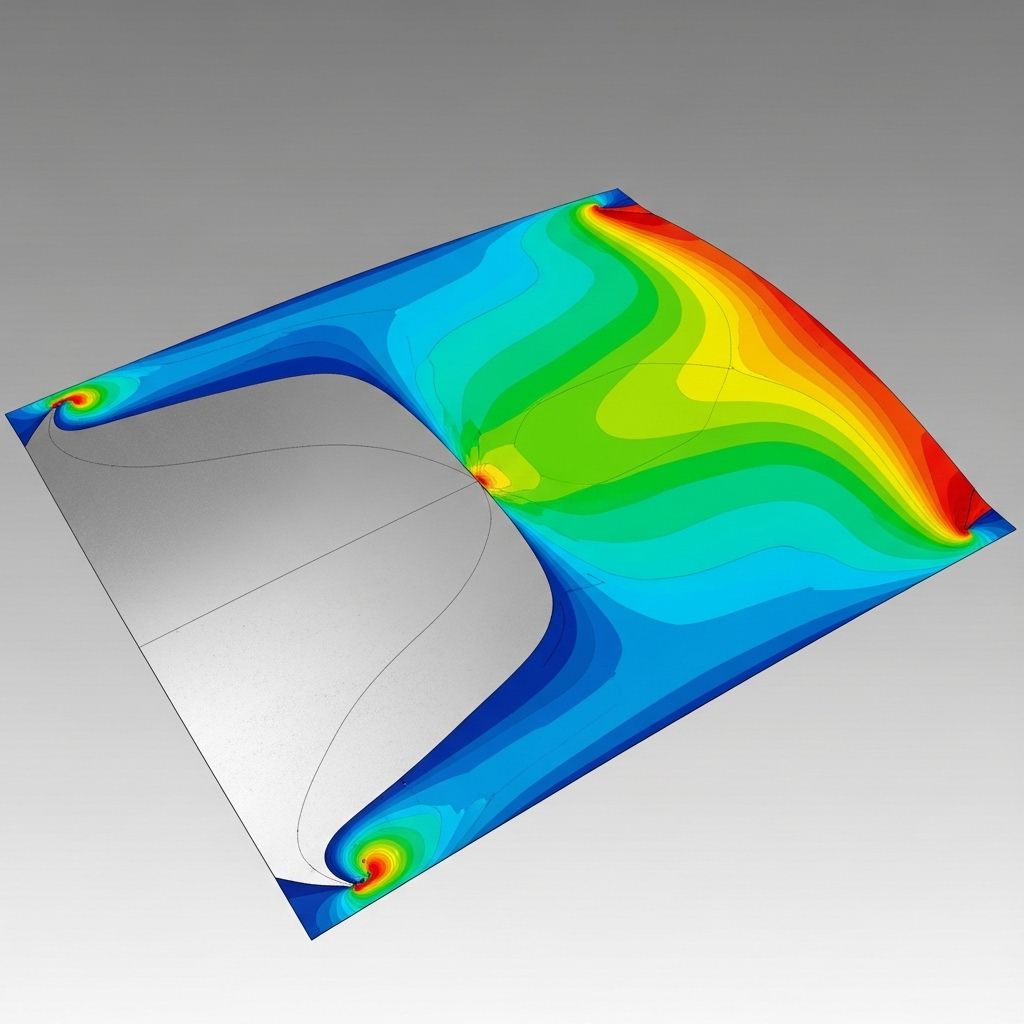
A detailed analysis of stress propagation, strain hardening, and the geometry of plastic deformation zones in sheet metal subjected to concentrated loads.
Read the full research
From hand-beaten coachwork to servo-controlled deep-draw stamping: a technical history of how automotive panels are engineered and produced.
Read the full researchCommon questions regarding the editorial approach, scientific methodology, and purpose of DentRepairHelp as an independent publisher.
DentRepairHelp is an independent digital publisher dedicated to the science of automotive metallurgy, structural materials physics, and molecular metal behaviour. This site exists to produce and disseminate rigorous, educational content for engineering enthusiasts, students, and professionals with an interest in how metals behave at the atomic and structural scale. We do not offer commercial services of any kind.
All articles are researched and drafted by specialist contributors with backgrounds in materials science, mechanical engineering, and industrial metallurgy. Content is reviewed against recognised standards from bodies including BSI, ASTM International, and ASM International before publication. Quantitative data presented in comparison tables is drawn from peer-reviewed materials databases and manufacturer technical datasheets.
No. DentRepairHelp does not sell products, provide cost estimates, generate referrals to commercial services, or accept compensation in exchange for editorial coverage. The site operates solely as an informational and educational publisher. Any display advertising that appears is managed through third-party networks and is editorially independent from site content.
Articles are written to be accessible to readers with a general interest in physics and engineering, while retaining technical rigour appropriate for those at undergraduate level or above. Where specialist terminology is used, explanatory context is always provided. The Metallurgy Glossary — accessible via the Articles section — provides definitions for all key terms used across the publication.
DentRepairHelp welcomes submissions from qualified contributors in the fields of materials science, mechanical engineering, and related disciplines. All submissions undergo editorial review for accuracy, originality, and alignment with the publication's focus on informational content. For full guest posting and editorial partnership guidelines, please visit the Partnership page.
Browse the complete catalogue of research articles, metal taxonomy guides, and physics explainers maintained by DentRepairHelp.
Read More Articles